Dans la même rubrique
-
Partager cette page
Epigenetic prognostic Biomarker of COVID-19 severity [Offre de technologie]
The technology in a nutshell
New prognostic Biomarker enabling early identification of patients infected by SARS (including COVID-19) at risk of developing severe illness, clinical complications and post-acute sequelae including pulmonary fibrosis.
State of the art
While efficient diagnostic methods have been developed to identify COVID-19 infection, biomarkers enabling early and accurate prediction of COVID-19 severity are still lacking. Diagnosis of COVID-19 severity is indeed still relying on clinical parameters and chest radiographic examination obtained on hospital admission. These stratification approaches, including changes in inflammatory markers such as leukocytes, proteins and metabolites, are time consuming and have strong limitations Rapid, economical and reliable quantitative methods enabling to accurately sort patients based on severity status and thereby provide an opportunity for better healthcare outcomes remains critical to optimize clinical COVID-19 patient triage.
The invention
The present invention is directed to the use of the fat mass and obesity associated protein (FTO) level of expression as biomarker for the prognosis of COVID-19 severity and corresponding stratification of patients. The FTO mRNA expression level measured by RNA sequencing or quantitative RT-PCR provides means to predict severity of the disease, to rapidly take therapeutic treatment decisions according to patient risk profile and to monitor clinical progression of the disease.
The technology based on a single biomarker is applicable to bronchioalveolar lavage samples of patients but also nasopharyngeal swabs and blood samples routinely used by physicians enabling thereby to optimize patient triage and resource allocation upon hospital admission.
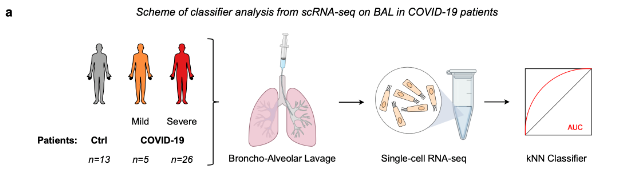
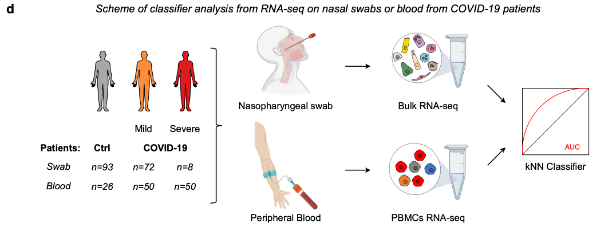
Figure - FTO expression in patient samples classifies COVID-19 severity.
a Classifier construction through k-Nearest Neighbor (kNN) machine learning on a single-cell RNA-seq dataset of gene expression levels in lung epithelial cells obtained from patient cohorts subjected to broncho-alveolar lavage (BAL), to nasal swab sampling or to blood sampling.
Key advantages of the technology
- Early diagnosis of disease severity upon hospital admission
- Efficient Covid-19 patient tirage based on routinely used sampling methods such as nasal swabs
- Method based on a single biomarker and readily available RT-PCR measurements
Potential applications
- Covid-19 patient stratification upon hospital admission for therapeutic treatment decision-making
- Monitoring of disease progression
Technology Readiness Level

TRL-3 Proof of concept assays established – Clinical validation ongoing.
The team
The Laboratory of Cancer Epigenetics led by François Fuks aims to investigate mechanisms underlying epigenetic modifications and how they go awry in several cancers and other diseases. Understanding the scope and mechanisms of dynamic RNA modifications, which is termed 'RNA epigenetics', is a main research focus of the laboratory. In this context the Laboratory of Cancer Epigenetics has addressed the distribution, function and biology of novel modifications of RNA such as RNA hydroxymethylation, opening thereby new research prospects in the emerging realm of RNA epigenetics. Besides studying fundamental mechanisms of DNA and RNA modifications, the laboratory has developed a growing interest in cancer epigenomics. In this respect, the laboratory is heading a state-of-the-art Next-Generation Sequencing core facility, dedicated to epigenetics. Regarding DNA methylome analyses, a thorough expertise has been developed in "Infinium Methylation" assays and in performing ChIP-Seq, RNA-Seq, ATAC-Seq, and their associated bioinformatics analyses.
The inventor

Relevant publication
Keywords
- Biomarker
- In Vitro Diagnosis/Prognosis
- COVID-19
- Disease Severity
Collaboration type
- Licence agreement
- R&D&I collaboration
IP status
-
EP22169473.0
Priority Date: 22/04/2022
Main inventor
François FUKS
Lionel MALBEC
Contact
ULB Research Department
Joachim Ruol
Business developer
+32 (0) 475 64 18 91
joachim.ruol@ulb.be
